DJO GLOBAL ANNOUNCES FINANCIAL RESULTS FOR SECOND QUARTER 2017
Business Transformation On Schedule
SAN DIEGO, CA, August 10, 2017 – DJO Global, Inc. (“DJO” or the “Company”), a leading global provider of medical technologies designed to get and keep people moving, today announced financial results for its public reporting subsidiary, DJO Finance LLC (“DJOFL”), for the second quarter ended July 1, 2017.
Second Quarter Financial Highlights
- Net sales grew 0.6% to $294.7 million, or 2.3% on a sales-per-day, constant currency basis.
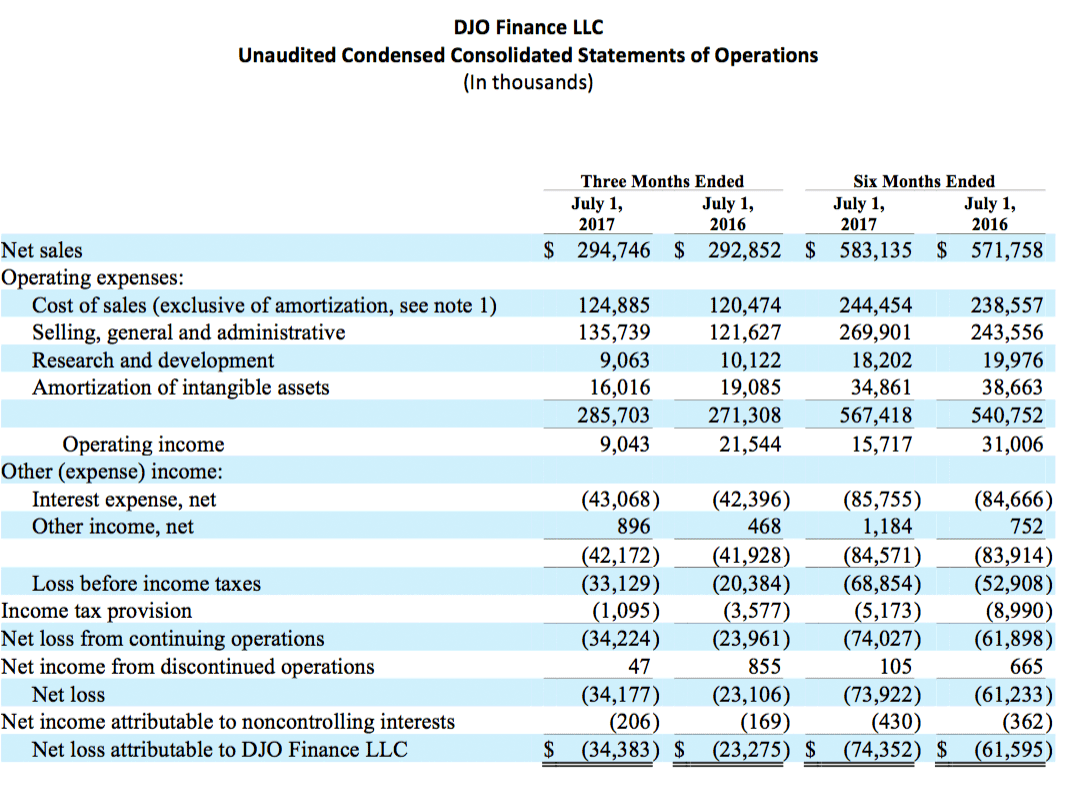
- Net loss attributable to DJOFL was $34.4 million compared to $23.3 million in the prior year period.
- Adjusted EBITDA was $63.5 million.
Business Transformation Progress
- Significant investments were made, as planned, in the Company’s transformation which remains on track to deliver 7% to 10% annual cost reduction by the end of 2018.
- Transformation actions taken to date expected to contribute $15 million in annual savings over the next four quarters.
“Our results for the second quarter, and for the first half, of the year are ahead of our annual operating plan and reflect the hard work our team has contributed to growing the business while transforming our operations,” said Brady Shirley, DJO’s President and Chief Executive Officer. “Over the first half of 2017, we continued to grow our global business and we grew Adjusted EBITDA faster than revenue. We also continued to execute on our business transformation, making investments and taking actions that support our priorities of improved profitability, liquidity, growth and customer experience. Looking forward, we remain confident in both our transformation and the team that we have in place to deliver long-term value to our customers, employees and investors.”
Sales Results
DJOFL achieved net sales for the second quarter of 2017 of $294.7 million, reflecting growth of 0.6%, compared with net sales of $292.9 million for the second quarter of 2016. The second quarter of 2017 included 61 shipping days for our international business compared to 63 for the same period in 2016, while domestic shipping days remained constant at 64 for both the second quarter of 2017 and 2016. Sales in the second quarter of 2017 grew 2.3% on a sales-per-day, constant currency basis over sales in the second quarter of 2016. For the six months ending July 1, 2017, sales grew 2.0% to $583.1 million over the same period in 2016, or 2.7% on a constant currency basis.
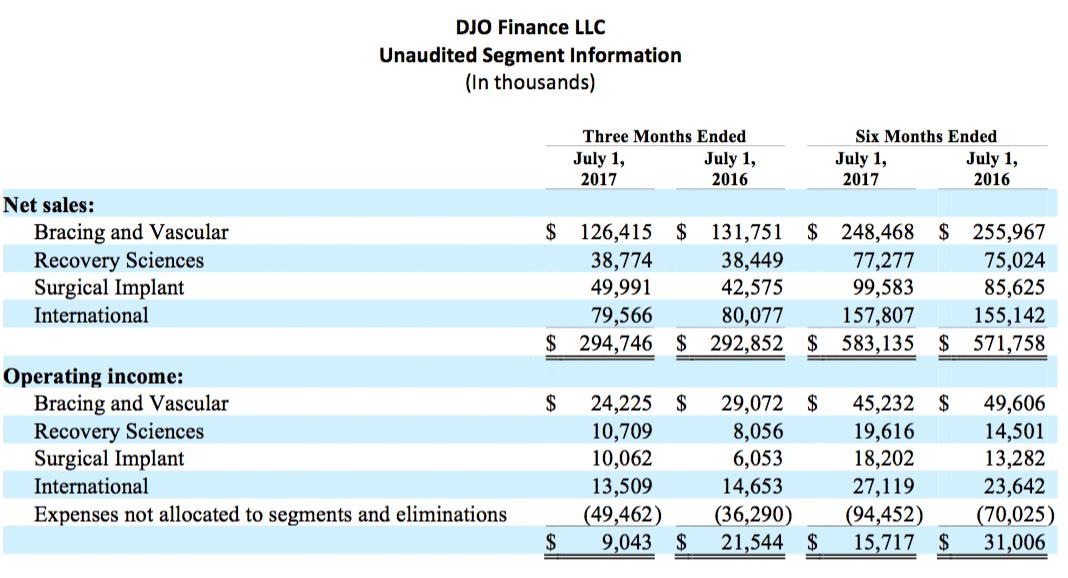
Net sales for the Surgical Implant segment grew 17.4% in the second quarter of 2017 to $50.0 million. Sales across all three implant subcategories (knee, hip and shoulder) again grew at double digit rates compared to the prior year. For the six months ending July 1, 2017, Surgical Implant sales grew 16.3%, over the comparable period in 2016, to $99.6 million.
Net sales for DJO’s International segment were $79.6 million in the second quarter of 2017, a decline of 0.6% compared to the second quarter of 2016. On the basis of constant currency and taking into account 61 shipping days for our international business compared to 63 in the prior year, International sales grew 5.3% driven by stronger sales in the Company’s direct markets, primarily Australia, France, and Spain, as well as continued growth in the Company’s export markets. For the six months ending July 1, 2017, International sales grew 1.7% to $157.8 million, or 4.3% on a constant currency basis over the comparable period in 2016.
Net sales for DJO’s Recovery Sciences segment were $38.8 million in the second quarter of 2017, reflecting growth of 0.8% compared to the second quarter of 2016. Growth in both of the segment’s major product lines, Chattanooga rehabilitation equipment and Regeneration CMF was relatively flat in the quarter compared to the prior year period. For the six months ending July 1, 2017, Recovery Sciences sales grew 3.0% to $77.3 million.
Net sales for DJO’s Bracing and Vascular segment were $126.4 million in the second quarter of 2017, a decline of 4.1%, compared to the second quarter of 2016, reflecting general softness across the Company’s bracing and support products, as well as continued pressure in the Company’s Dr. Comfort product line. For the six months ending July 1, 2017, Bracing and Vascular sales declined 2.9% to $248.5 million.
Earnings Results
For the second quarter of 2017, DJOFL reported a net loss of $34.4 million, compared to a net loss of $23.3 million for the second quarter of 2016. As detailed in the attached financial tables, the results for the current and prior year second quarter periods and the current and prior year twelve-month periods were impacted by significant non-cash items, non-recurring items and other adjustments.
Adjusted EBITDA for the second quarter of 2017 was $63.5 million compared with Adjusted EBITDA of $63.6 million in the second quarter of 2016. Adjusted EBITDA for the first six months of 2017 was $120.8 million compared with Adjusted EBITDA of $112.5 million in the first six months of 2016. Including projected future savings from cost savings programs currently underway of $15.0 million as permitted under our credit agreement and the indentures governing our outstanding notes, Adjusted EBITDA for the twelve months ended July 1, 2017 was $258.6 million.
The Company defines Adjusted EBITDA as net (loss) income attributable to DJOFL plus net interest expense, income tax provision (benefit), and depreciation and amortization, further adjusted for certain non-cash items, non-recurring items and other adjustment items as permitted in calculating covenant compliance under the Company’s secured term loan and revolving credit facilities (“Senior Secured Credit Facilities”) and the indentures governing its 8.125% second lien notes and its 10.75% third lien notes. A reconciliation between net loss attributable to DJOFL and Adjusted EBITDA is included in the attached financial tables.
Net cash provided by continuing operating activities for the six months ending July 1, 2017 was $38.1 million compared to a net use of cash of $10.6 million for the same period of 2016. The improvement in cash flow was primarily attributable to working capital initiatives executed as part of the Company’s overall business transformation.
Conference Call Information
DJO has scheduled a conference call to discuss this announcement beginning at 4:30 pm, Eastern Time Thursday, August 10, 2017. Individuals interested in listening to the conference call may do so by dialing (866) 394-8509 (International callers please use (706) 643-6833), using the reservation code 22322226. A telephone replay will be available for 48 hours following the conclusion of the call by dialing (855) 859-2056 and using the above reservation code. The live conference call and replay will be available via the Internet at www.DJOglobal.com.
About DJO Global
DJO Global is a leading global provider of medical technologies designed to get and keep people moving. The Company’s products address the continuum of patient care from injury prevention to rehabilitation, enabling people to regain or maintain their natural motion. Its products are used by orthopaedic surgeons, primary care physicians, pain management specialists, physical therapists, podiatrists, chiropractors, athletic trainers and other healthcare professionals. In addition, many of the Company’s medical devices and related accessories are used by athletes and patients for injury prevention and at-home physical therapy treatment. The Company’s product lines include rigid and soft orthopaedic bracing, hot and cold therapy, bone growth stimulators, vascular therapy systems and compression garments, therapeutic shoes and inserts, electrical stimulators used for pain management and physical therapy products. The Company’s surgical division offers a comprehensive suite of reconstructive joint products for the hip, knee and shoulder. DJO Global’s products are marketed under a portfolio of brands including Aircast®, Chattanooga, CMF™, Compex®, DonJoy®, ProCare®, DJO® Surgical, Dr. Comfort® and ExosTM. For additional information on the Company, please visit www.DJOglobal.com.
Safe Harbor Statement
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements relate to, among other things, the Company’s expectations for improved liquidity, estimated cost reductions associated with the execution of its business transformation plans and improved efficiencies. The words “believe,” “will,” “should,” “expect,” “target,” “intend,” “estimate” and “anticipate,” variations of such words and similar expressions identify forward-looking statements, but their absence does not mean that a statement is not a forward-looking statement. These forward-looking statements are based on the Company’s current expectations and are subject to a number of risks, uncertainties and assumptions, many of which are beyond the Company’s ability to control or predict. The Company undertakes no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise. The important factors that could cause actual operating results to differ significantly from those expressed or implied by such forward-looking statements include, but are not limited to the successful execution of the Company’s business transformation plans, including achievement of planned actions to improve liquidity, improvements in operational effectiveness, optimization of the Company’s procurement activities, improvements in manufacturing, distribution, sales and operations planning, and actions to improve the profitability of the mix of our product and customers. Other important factors that could cause actual operating results to differ significantly from those expressed or implied by such forward-looking statements include, but are not limited to: business strategies relative to our Bracing and Vascular, Recovery Sciences, International and Surgical Implant segments; the continued growth of the markets the Company addresses and any impact on these markets from changes in global economic conditions; the impact of potential reductions in reimbursement levels and coverage by Medicare and other governmental and commercial payors; the Company’s highly leveraged financial position; the Company’s ability to successfully develop, license or acquire, and timely introduce and market new products or product enhancements; risks relating to the Company’s international operations; resources needed and risks involved in complying with government regulations and government investigations; the availability and sufficiency of insurance coverage for pending and future product liability claims; and the effects of healthcare reform, Medicare competitive bidding, managed care and buying groups on the prices of the Company’s products. These and other risk factors related to DJO are detailed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2016, filed with the Securities and Exchange Commission on March 15, 2017. Many of the factors that will determine the outcome of the subject matter of this press release are beyond the Company’s ability to control or predict.
Note 1 — Cost of sales is exclusive of amortization of intangible assets of $6,980 and $13,961 for the three and six months ended July 1, 2017 and $7,080 and $14,487 for the three and six months ended July 1, 2016, respectively.
DJO Finance LLC
Adjusted EBITDA
For the Three and Six Months Ended July 1, 2017 and 2016
(unaudited)
Our Senior Secured Credit Facilities, consisting of a $1,036.5 million term loan facility (including a $20.0 million delayed draw term loan facility) and a $150.0 million asset-based revolving credit facility, under which $68.0 million was outstanding as of July 1, 2017, and the Indentures governing our $1,015.0 million of 8.125% second lien notes and $298.5 million of 10.75% third lien notes (collectively, the “notes”) represent significant components of our capital structure. Under our Senior Secured Credit Facilities, we are required to maintain a specified senior secured first lien leverage ratio, which is determined based on our Adjusted EBITDA. If we fail to comply with the senior secured first lien leverage ratio under our Senior Secured Credit Facilities, we would be in default. Upon the occurrence of an event of default under the Senior Secured Credit Facilities, the lenders could elect to declare all amounts outstanding under the Senior Secured Credit Facilities to be immediately due and payable and terminate all commitments to extend further credit. If we were unable to repay those amounts, the lenders under the Senior Secured Credit Facilities could proceed against the collateral granted to them to secure that indebtedness. We have pledged substantially all of our assets as collateral under the Senior Secured Credit Facilities and under the notes. Any acceleration under the Senior Secured Credit Facilities would also result in a default under the Indentures governing the notes, which could lead to the note holders electing to declare the principal, premium, if any, and interest on the then outstanding notes immediately due and payable. In addition, under the Indentures governing the notes, our and our subsidiaries’ ability to engage in activities such as incurring additional indebtedness, making investments, refinancing subordinated indebtedness, paying dividends and entering into certain merger transactions is governed, in part, by our ability to satisfy tests based on Adjusted EBITDA. Our ability to meet the covenants specified in the Senior Secured Credit Facilities and the Indentures governing those notes will depend on future events, some of which are beyond our control, and we cannot assure you that we will meet those covenants.
Adjusted EBITDA is defined as net income (loss) attributable to DJOFL plus interest expense, net, income tax provision (benefit), and depreciation and amortization, further adjusted for certain non-cash items, non-recurring items and other adjustment items as permitted in calculating covenant compliance and other ratios under our Senior Secured Credit Facilities and the Indentures governing the notes. We believe that the presentation of Adjusted EBITDA is appropriate to provide additional information to investors about the calculation of, and compliance with, certain financial covenants and other ratios in our Senior Secured Credit Facilities and the Indentures governing the notes. Adjusted EBITDA is a material component of these calculations.
Adjusted EBITDA should not be considered as an alternative to net income (loss) attributable to DJOFL or other performance measures presented in accordance with accounting principles generally accepted in the United States of America (“GAAP”), or as an alternative to cash flow from operations as a measure of our liquidity. Adjusted EBITDA does not represent net income (loss) attributable to DJOFL or cash flow from operations as those terms are defined by GAAP and does not necessarily indicate whether cash flows will be sufficient to fund cash needs. In particular, the definition of Adjusted EBITDA under our Senior Secured Credit Facilities and the Indentures governing the notes allows us to add back certain non-cash, extraordinary, unusual or non-recurring charges that are deducted in calculating net income (loss) attributable to DJOFL. However, these are expenses that may recur, vary greatly and are difficult to predict. While Adjusted EBITDA and similar measures are frequently used as measures of operations and the ability to meet debt service requirements, Adjusted EBITDA is not necessarily comparable to other similarly titled captions of other companies due to the potential inconsistencies in the method of calculation
The following table provides reconciliation between net loss attributable to DJOFL and Adjusted EBITDA (in thousands):
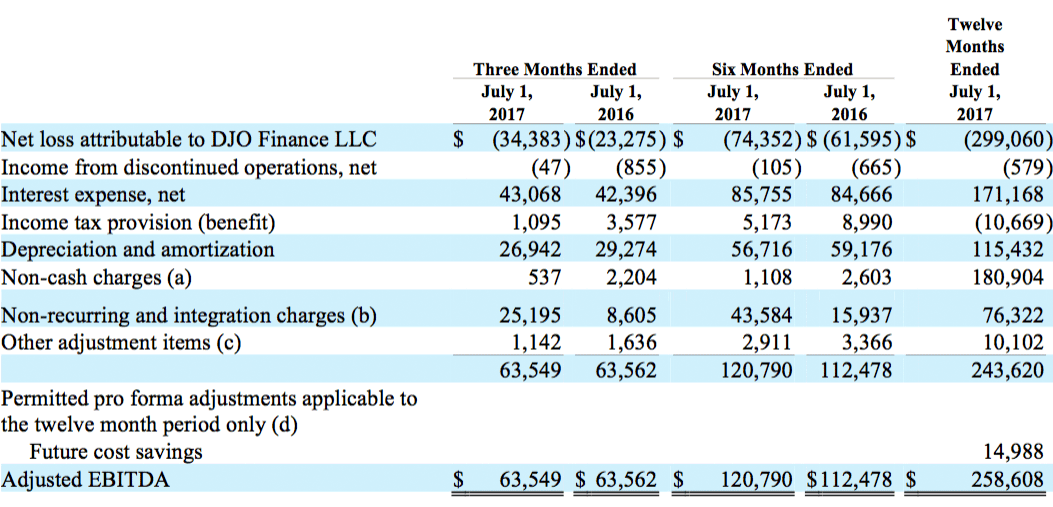
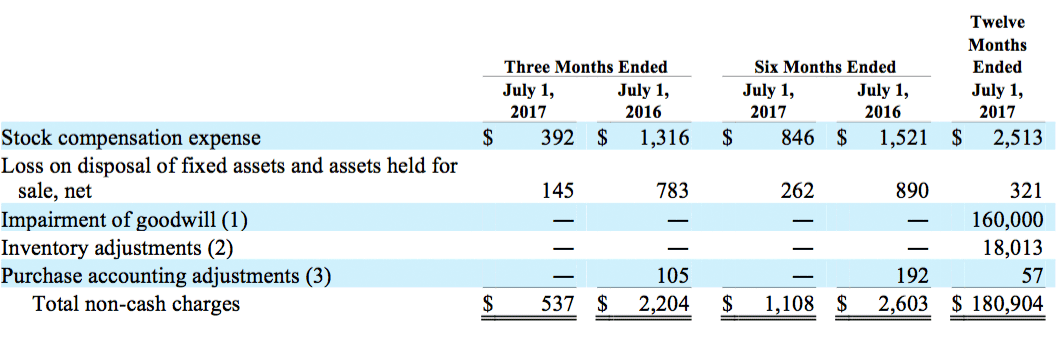
(1) Impairment of goodwill and intangible assets for the twelve months ended July 1, 2017 consisted of goodwill impairment charges of $99.0 million and $61.0 million related to the CMF and Vascular reporting units, respectively. The impairment charge for our CMF reporting unit resulted from reductions in our projected operating results and estimated future cash flows due to disruption caused by our exit of the Empi business. The impairment charge for our Vascular reporting unit resulted from reductions in our projected operating results and estimated future cash flows due to a loss of revenue caused by disruption as we transitioned our Dr. Comfort therapeutic footwear manufacturing and distribution to a new ERP system and market pressure in the therapeutic shoe market.
(2) In the fourth quarter of fiscal 2016, current management implemented a new strategy relating to our procurement, manufacturing and liquidation philosophies in order to significantly reduce inventory levels. Historically, our strategy was to purchase inventory in large quantities to capture purchase discounts and rebates and provide an expansive mix of products for our customers. Our new strategy aims to integrate our supply chain services with customer demand through focused forecasted consumption and sales efforts, therefore limiting the range of SKUs we plan to offer. As a result of these changes, the Company recorded a charge to cost of sales and corresponding reduction in inventory of approximately $18.0 million. The E&O reserve expense in fiscal 2016 included $5.7 million related to the Company’s decision to discontinue certain SKUs mainly within the Bracing and Vascular product lines, $8.3 million related to holding inventory for shorter periods and the planned scrapping of long-dated inventory, $2.0 million related to new Surgical Implant products that changed the expected life cycle of its current product portfolio, and $2.0 million of slow moving consigned inventory within certain OfficeCare clinics for which management has decided not to strategically relocate.
(3) Purchase accounting adjustments consisted of amortization of fair market value inventory adjustments for all periods presented.
(b) Non-recurring and integration charges are comprised of the following:
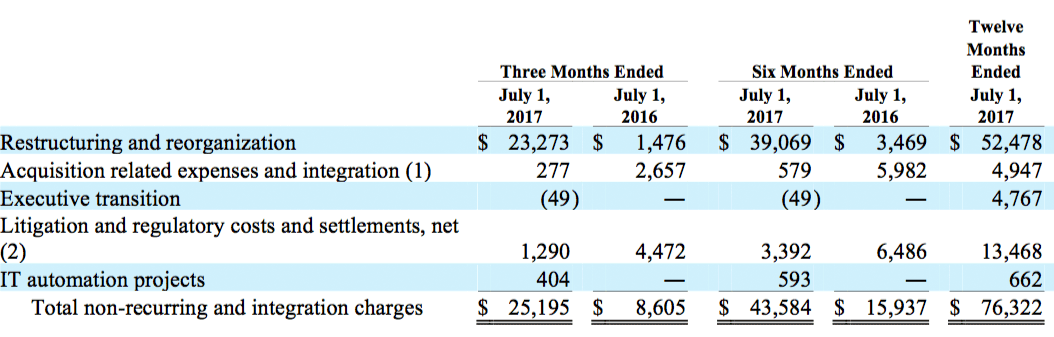
(1) Consists of direct acquisition costs and integration expenses related to acquired businesses and costs related to potential acquisitions.
(2) For the twelve months ended July 1, 2017, litigation and regulatory costs consisted of $1.4 million in litigation costs related to ongoing product liability issues and $12.1 million related to other litigation and regulatory costs and settlements.
(c) Other adjustment items are comprised of the following:
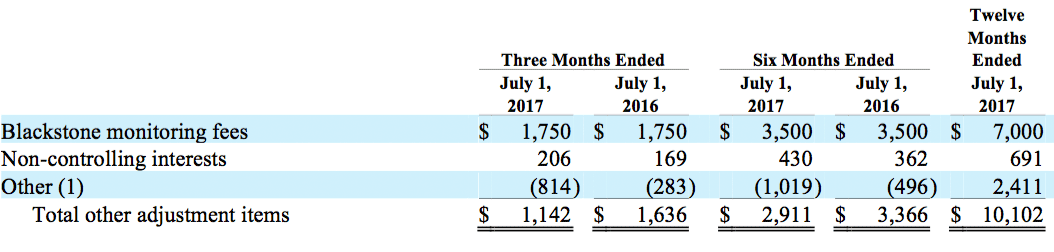
(1) Other adjustments consist primarily of net realized and unrealized foreign currency transaction gains and losses.
(d) Permitted pro forma adjustments include future cost savings related to the exit of our Empi business and our business transformation initiative.
